Detecting Aphid Concentration in Wheat Leaf Using Remote Sensing and GIS
Rao Mansor Ali Khan1, Saira Batool Naqvi1, Qamar Shabbir Rana1, Syed Amer Mahmood1, Mamoon ur Rasheed1
1 Department of Space Science, University of Punjab.
* Correspondence: Rao Mansor Ali Khan, Email ID: Raomansor@gmail.com
Citation | Khan. R. M. A, Naqvi. S. B, Rana. Q. S, Mahmood. S. A, Rasheed. M, “Detecting Aphid Concentration in Wheat Leaf Using Remote Sensing and GIS”. International Journal of Innovations in Science and Technology, Vol 4, Issue 2, 2022, pp: 336-347.
Received | April 5, 2022; Revised | April 19, 2022 Accepted | April 19, 2022; Published | April 20, 2022.
________________________________________________________________________
Wheat lies among the most prominent cereal crop of Pakistan which has a significant role in the stability of Pakistan’s economy. Certain biotic and a biotic factors including agro-climatic conditions, rainfall, lack of irrigation infrastructure and conventional agricultural methods are raising the risk of aphid attacks. The current study utilized satellite imagery for obtaining thermal datasets of complete wheat growth. Results revealed that rainfall is a significant parameter for the determination of aphid growth on wheat plant. A region receiving 0-10 mm rainfall, supported the growth of aphid. Moreover, the aphid survival was highly supported at a moderate temperature ranging between 20-25 oC with relative humidity ranging from 70-75 %. The study also revealed the production of weed in wheat crop acted as a moistrizing agent which consequently provided favorable conditions to the aphid population for growth. Inapropriate usage of fertilizers increased the nitrogen content in soil which turned to be favourable for the aphid attack. Thus, the study concludes that agro-climatic conditions must be considered before the aplication of pesticides.
Key Words: Aphid; Conventional Agricultural method; Moistourizing agent; Agro-climatic condition.
|
Author’s Contribution All authors contributed equally. |
CONFLICT OF INTEREST: The author(s) declare that the publication of this article has no conflict of interest. |
Project details. Nil
|
Introduction
Wheat (Triticum aestivum L.) is a prominent crop in Pakistan[1], with the largest area under cultivation and plays a crucial part in the country's economic stability [2]. Traditional methods of agriculture, lack of irrigation infrastructure, barani regions, soil fertility, and the insect pest attacks are the major factors responsible for a decline in wheat production which ultimately effects the regional economy in comparison to other neighboring countries. Besides, cultural, physical, mechanical, biological, chemical aspects, host plant resistance is also considerable parameter which may not be ignored while evaluating the overall production. Aphid has been observed one of the most threatening and a yield degrading factor among others. Aphids reduce yields either directly (35-40%) by sucking the plant's sap or indirectly (20-80%) by transferring viral and fungal infections [3].
Abiotic factors are also responsible for an increase in Aphid population [4]–[6]. The population of aphids increases throughout the spring season[7] (February-March), and biocontrol agents such as coccinellids grow as a natural check on this pest [8]. A combination of naturally occurring population controlling variables keeps aphid populations below the economic harm threshold. However, if aphids are present in great numbers, they can be extremely harmful, necessitating the application of insecticides to control them [9]. The wheat crop is commonly plagued with aphids during the growth stages, when both adults and nymphs wreak havoc on the plants by sucking cell sap and reducing the plant’s vitality [10][11]. The infested leaves become paler, wilt, and seem silky[12]. Toxins are found in the saliva of several species, and a dense infestation can destroy new shoots[13]. Aphid infestations are commonly accompanied by honeydew excretion and sooty molds, both of which affect the rate of photosynthesis in plants[14]. The wheat crop's low yield is primarily due to its vulnerability to aphid infestation [15]. Aphids are regarded as a major pest of wheat crops[16]. Under ideal conditions, they can quickly multiply on leaves, stems, and inflorescence[17]. The infestation produces severe leaf and inflorescence deformation, as well as a considerable reduction in yield due to direct feeding. Abiotic factors such as conventional methods of farming, poor yielding cultivars, and a lack of sufficient irrigation facilities in most places are all contributing to the drop in wheat crop output.
The population of Aphid is highly dependent upon agro-climatic conditions [18] and a rapid shift has been observed in the whole weather system e.g., the rainy season have been shifted and we receive rainfall at inappropriate times/ places. Our farmer religiously follow the crop calendar where the dates/ time of every event is fixed therefore, the application of pesticides and insecticides at unsuitable times cause a lost to local economy which is ultimately contributing in regional financial affairs. Therefore, it is a need of the hour to create an awareness among individuals to adopt the latest technologies/ farming practices for sustainable/ economic agriculture.
The main objective of this research is to delineate the vulnerable zones to Aphid incorporating regional environmental parameters including temperature, humidity and rainfall. It also aim to take remedial measures in time/ space to ensure the wheat growth and development in a protective environment.
Material and Methods
In the current section, the material and methodology adopted in this research is described in details. The data used in this research include thermal datasets based on satellite imagery for the complete growth period of wheat crop. Moreover, the humidity data was recorded through hygrometer and rainfall data, which was collected from Pakistan Metrological Department (PMD). The step-by-step methodology adopted in this research is as below
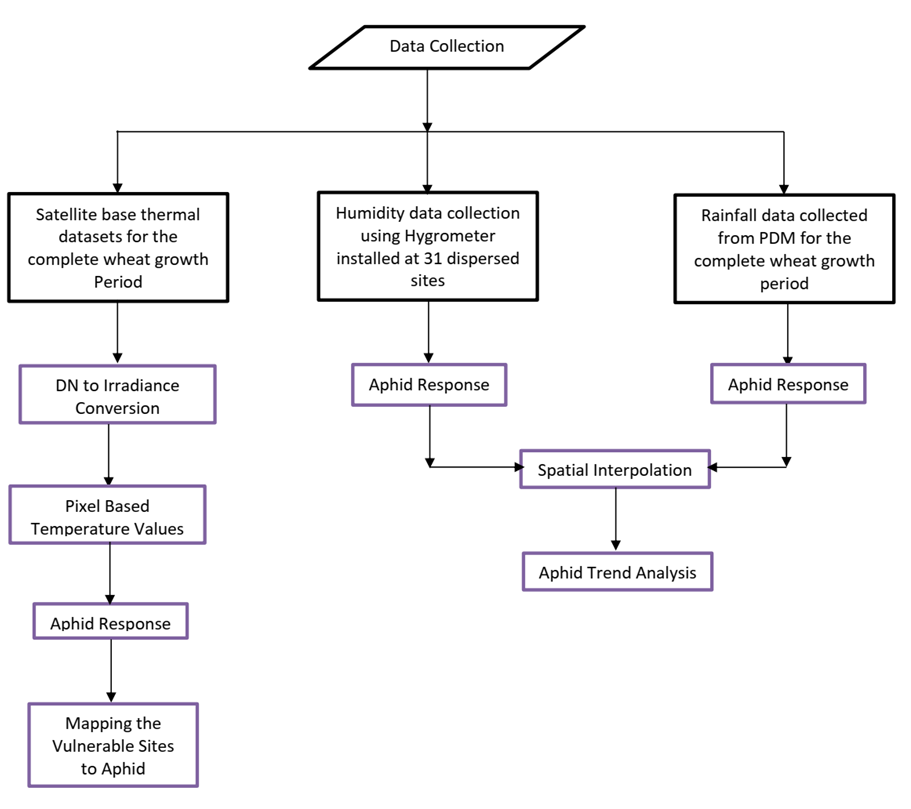
Figure 1. Flow of methodology
Aphid is an insect of wheat crop which is considered as the largest limiting factor to crop productivity[19]. In this research, various kinds of datasets including rainfall, temperature and humidity were utilized to determine the spatial distribution of aphid dynamics in the wheat crop. Aphid is very sensitive to humidity and its population get many folds as the humidity gets double[20]. To check the trend of humidity, we installed hygrometers at 31x dispersed spatial locations in the study site. The data was collected against each growth stage of wheat crop therefore about 160 values (for each location) of humidity were recorded during the complete growth/development period. These values were averaged and the interpolation technique IDW was applied on 31 locations to map the trend of humidity as shown in Figure 2 as below,
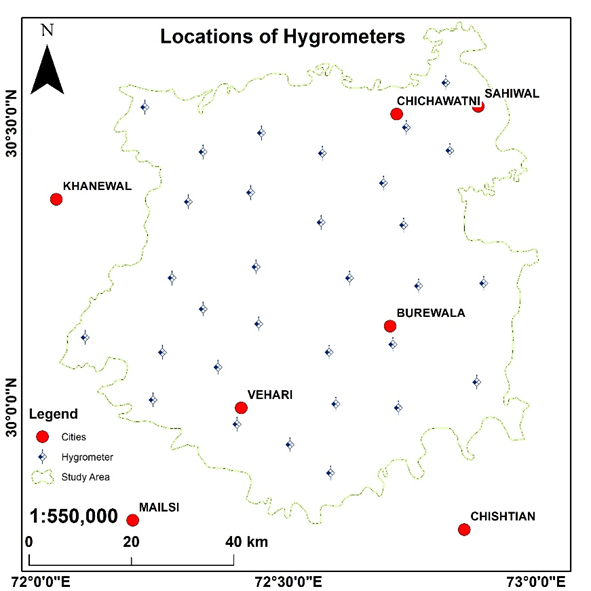
Figure 2. Spatial distribution of Hygrometers installed in the study site
Pakistan Meteorological Department (PMD) is continuously collecting the rainfall data in mm. Rainfall spells received during the complete study span in the investigations site is shown in Table 1.
Table 1. Rainfall Spells during Aphid life cycle.
|
Sr No |
Month |
Rainfall Spells |
|
1. |
February |
04-05 Feb 2020 |
|
2. |
12-14 Feb 2020 |
|
|
3. |
20-21 Feb 2020 |
|
|
4. |
28-29 Feb 2020 |
|
|
5. |
March |
05-08 Mar 2020 |
|
6. |
12-14 Mar 2020 |
|
|
7. |
21-24 Mar 2020 |
|
|
8. |
26-27 Mar 2020 |
|
|
9. |
April |
01-10 April 2020 |
|
10. |
11-20 April 2020 |
|
|
11. |
21-30 April 2020 |
PMD generated rainfall maps which were scanned, geo-referenced and digitized to extract actionable information regarding spatial distribution of rain.
Landsat 8 datasets are comprised of two thermal bands including Band 10 and Band 11[21] which provide very precise and reliable information regarding spatial distribution of temperature[22]. We downloaded these thermal bands for the complete wheat growth period and computed pixel-based temperature values. The information regarding downloaded datasets is as below,
Table 2. Rainfall Spells during Aphid life cycle.
|
Sr No |
Growth Stage of Wheat Crop |
Date of imagery |
|
3. |
Heading |
06 Feb 2020 |
|
4. |
Milky Dough |
09 Mar 2020 |
|
5. |
Maturity |
10 April 2020 |
Thermal bands are comprised of array of brightness values which are converted to irradiance [23]using the equations as below,
Irradiance B10= 0.0003342 * B10 + 0.1 (1)
Irradiance B11=0.0003342 * B11 + 0.1 (2)
The irradiance is further converted to temperature values using the equations[24] as below,
T B10 = {K2/ Ln (Ɛ K1 /Irradiance B10 + 1)} – 272.15 (3)
T B11= {K2/ Ln (Ɛ K1 /Irradiance B11 + 1)} – 272.15 (4)
In equations (3) and (4), K1 and K2 are the constants for thermal bands and their values are mentioned in the metadata of Landsat 8 [25] which are;
K1 for B10 = 774.89; K1 for B11 = 480.89; K2 for B10 = 1321.08; K2 for B11 = 1201.14
Value of emissivity (Ɛ) used in the equations (3) and (4) is 0.95. Final temperature against each pixel is computed by taking an average of both datasets T B10 and T B11.
T = (T B10 + T B11) / 2
Pixel based temperature values are computed using “Raster Calculator”, an elegant tool embedded in Arc GIS 10.1.
Result and discussions.
Temperature based vulnerable zones for Aphid.
Spatial distribution of temperature was computed using thermal datasets enlisted in Table 2. The stages of wheat crop affected by Aphid has been incorporated and categorized into less, moderate and not favorable zones. The basic reason for computing the pixel-based temperature values only for the month of Feb, March and April is that Aphid emerges in the month of Feb and the complete life cycle ends up to April. Spatial distribution of temperature across the study site is mapped in Figure 2.

Figure 3. Spatial distribution of vulnerable zones for Aphid during months Feb, March and April.
The results are given in Table below
Table 3. Areas in km2 regarding vulnerable zones to Aphid.
|
Sr No |
Date of Satellite Imagery |
Favorable area km2 |
Moderately area km2 |
Not Favorbale area km2 |
|
1. |
06 Feb 2020 |
73 |
3987 |
1638 |
|
2. |
09 Mar 2020 |
129 |
4123 |
1704 |
|
3. |
10 April 2020 |
153 |
4568 |
1283 |
Rainfall based vulnerable zones for Aphid.
Rainfall acts as a promoting factor for growth in population of Aphid. It has been observed that just after 01 x day of rainfall, the insects come out of their holes and the population increases many folds. We could get 11 x spells of rainfall during Feb-April. The intensity of rainfall was geotagged and interpolated. The results of interpolation are mapped in Figure 3-5.
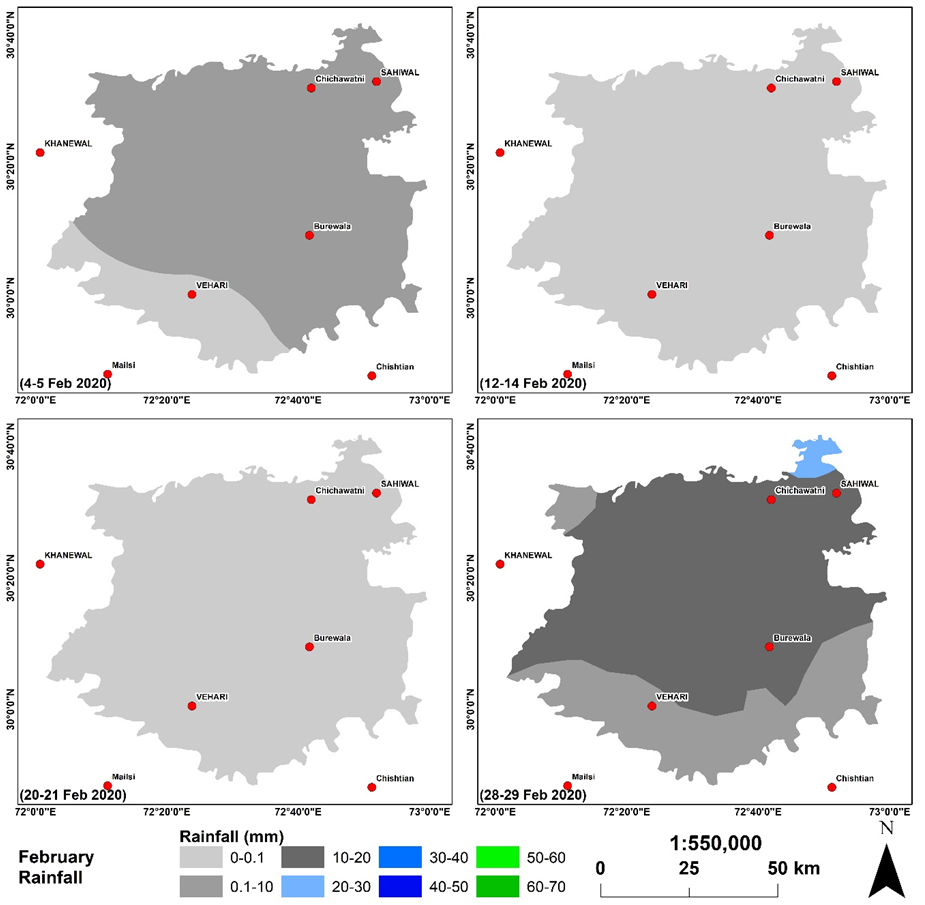
Figure 4. Spatial distribution of rainfall intensity during the month of Feb 2020.
|
Figure 5. Spatial distribution of rainfall intensity during the month of March 2020. |
|
Figure is showing the spatial distribution of rainfall across the study site during the month of Feb 2020. Most of rainfall received in the fourth spell from 28-29 Feb 2020 in the northeast with rainfall intensity 20-30 mm. The effect of rainfall in other spells did not effect the Aphid growth however, in the forth spell, the dark grey and light blue areas are showing instable zone for Aphid to stay on the plant and they fall on the ground. Spell 2 and 3 are showing the rainfall intensity less than 10 mm that did not effect the the Aphid existance therefore, its population increased exponetially untill the fouth spell hit the area. It takes about 1 week for Aphid to reinstate into its previous condition of the wheat plant which was observed before the first spell of march that receieved heavy rainfall in specific regions highlighted with green and blue colors which were observed the death regions for Aphid however, the blue regions receieved 40-50 mm which was so intense that created danger for Aphid survival. During march 2020, the study site receieved more intense rainfall as comparted to the previous month. The intensity of rainfall was not homogenous but it was diversed therefore the behaviour of Aphid was not similar throughout the study site.
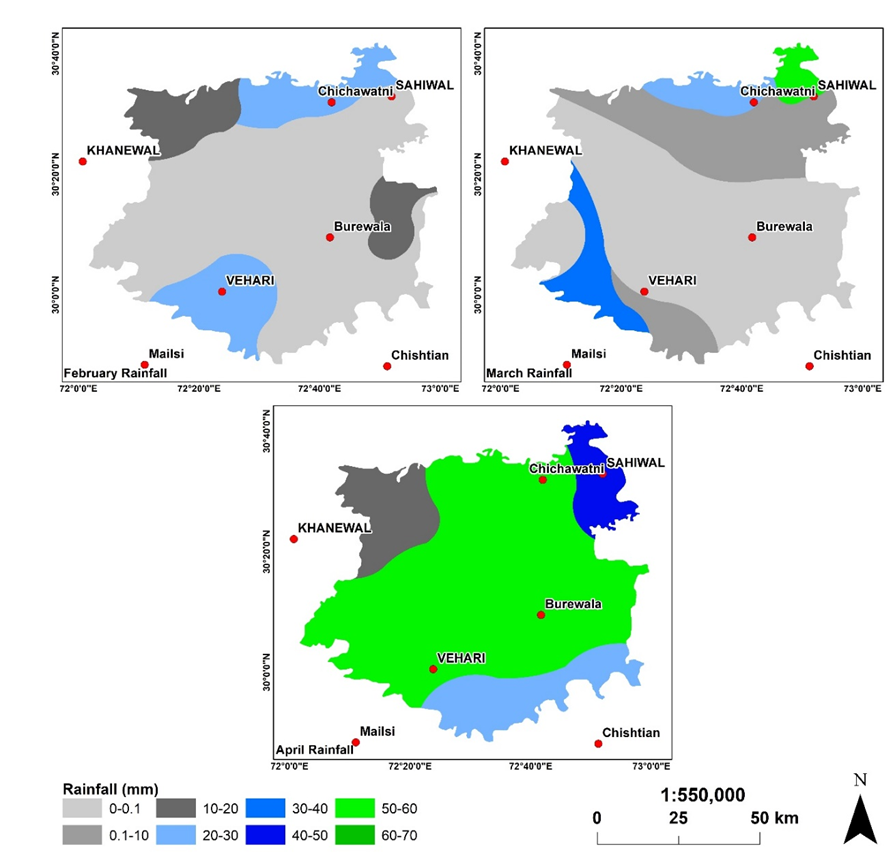
Figure 7. Integarted map of rainfall.
Rainfall is an important factor to determine the population of Aphid. Aphid grow in the regions receiving rainfall within a range of 0-10 mm and if the rainfall is greater than 10mm, aphid destablizes and can not stay on the wheat stem/ leaf. Thefore, healthy rainfall is considered favorable to escape form aphid however, cloudy conditions for long times result to boost aphid population.
Humidity based vulnerable zones for Aphid.
Humidity plays a vital role toward the population of Aphid. As humidity increases, the population increases exponentially. The spatial distribution of humidity as recorded by 31 well distributed sites is mapped in Figure 8. Humidity maps were generated of each month from Feb- April to evaluate the existence/vulnerable zones for Aphid existence.
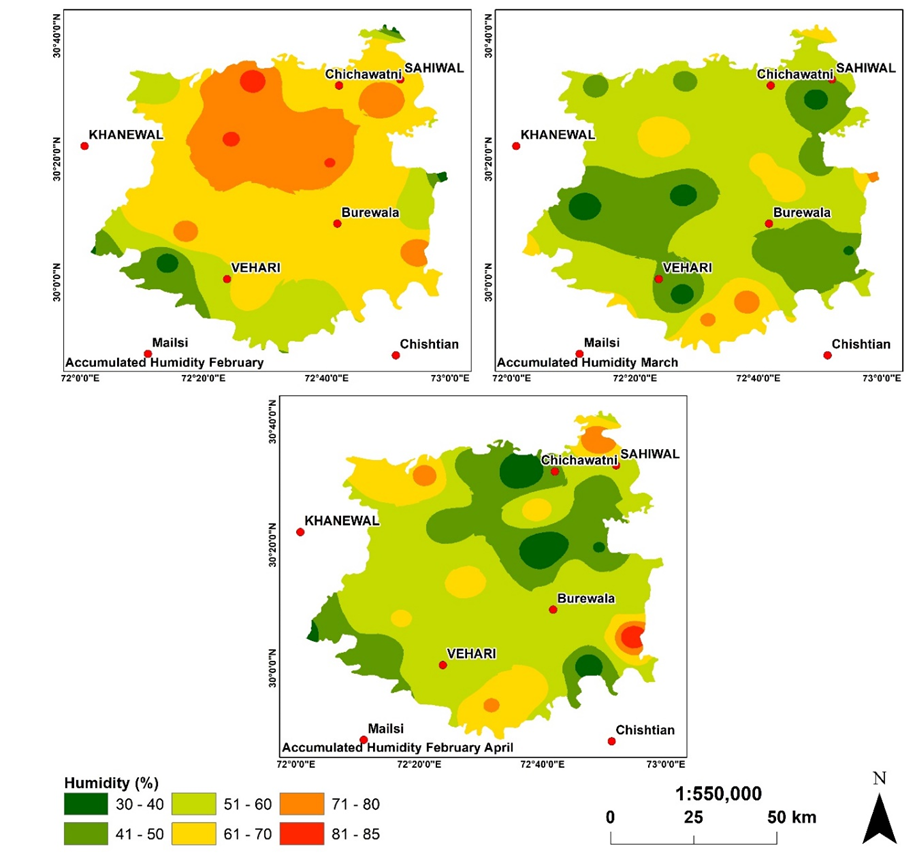
Figure 8. Humidity maps were generated of each month from Feb- April.
Discussions.
The results determine that moderate temperature of 20-25 oC with relative humidity ranging from 70-75 % is highly suitable for aphid to survive therefore, these climatic conditions may be treated to apply pesticides on emergency grounds. It has been observed that weed act as moisturizing agent and provide favoroable conditions for aphid to have proper growth therefore weeds must be abolished to remove safe houses for aphid.
Sowing time of wheat crop play a significant role against attacks/damages by aphid. It is recommended that wheat sowing practices must be completed before 25 Nov which results in hard dough that provides a shield against aphid attacks. The inapropriate applications of fertilizers invite aphid to attack the wheat crop, e.g., the excessive use of nitrogen in comparison to phosphorus results in high population of aphid. Number of irrigations also effects the aphid population. If there exist sufficient moisture content in wheat field. There are healthy chances of aphid to damage the wheat crop.
Adoption of sowing strategy is also important to control the aphid population. In case of uneven distribution of wheat seeds, the wind is unable to pass through the wheat plants which provide favourable zone to aphid for growth and development. Moreover, 30kg/acre is recommended at sowing time that results in sparse plantation however, 50kg/acre results in dense wheat crop which is favourable for aphid to survive for long times.
References
[1] J. Anwar et al., “Assessment of yield criteria in bread wheat through correlation and path analysis,” J. Anim. Plant Sci., vol. 19, no. 4, pp. 185–188, 2009.
[2] M. Aslam, M. Razaq, W. Akhter, M. Faheem, and F. Ahmad, “Effect of sowing date of wheat on aphid (Schizaphis gramium RONDANI) population,” Pakistan Entomol., no. May, 2005.
[3] Aheer GM, Munir M, Ali A (2007) Impact of weather factors on population of wheat aphids at Mandi Baha-ud-Din district. J Agric Res 45: 61–66. .
[4] H. Jan et al., “Effect of biotic and abiotic factors on the population dynamics of wheat aphids,” vol. 5, no. 6, pp. 2349–2352, 2017.
[5] Z. ABBASI, R. SULTANA, M. S. WAGAN, and F. SOOMRO, “Population, Fluctuation of Aphid on the different Wheat Varieties from Larkana District,” Sindh Univ. Res. J. -Science Ser., vol. 51, no. 03, pp. 411--418, 2019, doi: 10.26692/surj/2019.09.66.
[6] A. A. Khan et al., “Effect of wheat cultivars on aphids and their predator populations,” African J. Biotechnol., vol. 10, no. 80, pp. 18399–18402, 2011, doi: 10.5897/AJB11.2961.
[7] J. H. Hatchett, K. J. Starks, and J. A. Webster, “Insect and Mite Pests of Wheat,” Wheat Wheat Improv., vol. 13, pp. 625–675, Oct. 2015, doi: 10.2134/AGRONMONOGR13.2ED.C34.
[8] “An ‘agricultural’ Pakistan — a necessity » Pakissan.com.” https://www.pakissan.com/2018/12/07/an-agricultural-pakistan-a-necessity/ (accessed Apr. 11, 2022).
[9] “Pakistan: Grain and Feed Update | USDA Foreign Agricultural Service.” https://www.fas.usda.gov/data/pakistan-grain-and-feed-update-18 (accessed Apr. 11, 2022).
[10] A. K. Muhammad, “Wheat Crop Yield Losses Caused by the Aphids Infestation,” J. Biofertilizers Biopestic., vol. 03, no. 04, pp. 10–12, 2012, doi: 10.4172/2155-6202.1000122.
[11] A. Kibe, S. Singh, and N. Kalra, “Water-nitrogen relationships for wheat growth and productivity in late sown conditions,” J. Agric. Sci. Technol., vol. 7, no. 1, pp. 1–17, May 2006, doi: 10.4314/jagst.v7i1.31716.
[12] R. A. Memon, G. R. Bhatti, S. Khalid, and S. Ahmed, “Illustrated weed flora of cotton crop of Khairpur district, Sindh, Pakistan,” Pakistan J. Bot., vol. 46, no. 1, pp. 5–12, 2014.
[13] S. A. Ba-Angood and R. K. Stewart, “EFFECT OF CEREAL APHID INFESTATION ON GRAIN YIELD AND PERCENTAGE PROTEIN OF BARLEY, WHEAT, AND OATS IN SOUTHWESTERN QUEBEC,” Can. Entomol., vol. 112, no. 7, pp. 681–686, Jul. 1980, doi: 10.4039/ENT112681-7.
[14] M. Hullé, B. Chaubet, E. Turpeau, and J. C. Simon, “Encyclop’aphid: A website on aphids and their natural enemies,” Entomol. Gen., vol. 40, no. 1, pp. 97–101, 2020, doi: 10.1127/entomologia/2019/0867.
[15] J. Lenné, “Pests and poverty: The continuing need for crop protection research,” Outlook Agric., vol. 29, no. 4, pp. 235–250, 2000, doi: 10.5367/000000000101293301.
[16] X. F. Liu, X. S. Hu, M. A. Keller, H. Y. Zhao, Y. F. Wu, and T. X. Liu, “Tripartite Interactions of Barley Yellow Dwarf Virus, Sitobion avenae and Wheat Varieties,” PLoS One, vol. 9, no. 9, p. e106639, Sep. 2014, doi: 10.1371/JOURNAL.PONE.0106639.
[17] . S. M. K. and . R. M., “Varietal Performance of Wheat (Triticum aestivum) Against Wheat Aphid (Macrosiphum miscanthi) and its Chemical Control with Different Doses of Insecticides,” Asian J. Plant Sci., vol. 1, no. 2, pp. 205–207, Feb. 2002, doi: 10.3923/AJPS.2002.205.207.
[18] R. Mahmood, M. A. Poswal, and A. Shehzad, “Distribution, Host Range and Seasonal Abundance of Sipha Sp. (Homoptera: Aphididae) and Their Natural Enemies in Pakistan,” Pakistan J. Biol. Sci., vol. 5, no. 1, pp. 47–50, Dec. 2001, doi: 10.3923/PJBS.2002.47.50.
[19] J. Liu, Y. Yan, A. Ali, N. Wang, Z. Zhao, and M. Yu, “Effects of wheat-maize intercropping on population dynamics of wheat aphids and their natural enemies,” Sustain., vol. 9, no. 8, 2017, doi: 10.3390/su9081390.
[20] M. Razaq and F. Ahmad, “Population of aphid (Schizaphis graminum R.) on different varieties/lines ofwheat (Triticum aestivum L.),” Int. J. Agric. Biol., vol. 6, no. 6, pp. 974–977, 2004.
[21] T. Perveen Inayat, S. Akhtar Rana, N. Rana, T. Ruby, M. Javaid Iqbal Sadiqui, and M. Nadeem Abbas, “Predation rate in selected coccinellid (coleoptera) predators on some major aphidid and cicadellid (hemipteran) pests,” Int. J. Agric. Biol., vol. 13, no. January, pp. 427–430, 2011, [Online]. Available: http://www.fspublishers.org.
[22] D. H. Kazmi, G. Rasul, D. H. Kazmi, and G. Rasul, “Agrometeorological wheat yield prediction in rainfed Potohar region of Pakistan,” Agric. Sci., vol. 3, no. 2, pp. 170–177, Mar. 2012, doi: 10.4236/AS.2012.32019.
[23] M. K. Khattak, Riazuddin, and M. Anayatullah, “Population dynamics of aphids (Aphididae: Homoptera) on different wheat cultivars and response of cultivars to aphids in respect of yield and yield related parameters,” Pak. J. Zool., vol. 39, no. 2, pp. 109–115, 2007.
[24] S. Saleem, F. Ullah, and M. Ashfaque, “Population dynamics and natural enemies of aphids on winter wheat in Peshawar, Pakistan,” Pak. J. Zool., vol. 41, no. 6, pp. 505–513, 2009.
[25] Q. Zeb, H. Badshah, H. Ali, R. Shah, and M. Rehman, “Population of aphids on different varieties/lines of wheat and their effect on yield and thousands grain weight,” Sarhad J. Agric, vol. 27, no. 3, pp. 443–450, 2011, [Online]. Available: http://www.aup.edu.pk/sj_pdf/population on aphid.PDF.
[26] J. A. MANN, G. M. TATCHELL, M. J. DUPUCH, R. HARRINGTON, S. J. CLARK, and H. A. McCARTNEY, “Movement of apterous Sitobion avenae (Homoptera: Aphididae) in response to leaf disturbances caused by wind and rain,” Ann. Appl. Biol., vol. 126, no. 3, pp. 417–427, 1995, doi: 10.1111/J.1744-7348.1995.TB05376.X.
[27] N. Elliott and R. W. Kieckhefer, “Response by coccinellids to spatial variation in cereal aphid density,” Popul. Ecol. 2000 421, vol. 42, no. 1, pp. 81–90, 2000, doi: 10.1007/S101440050012.
[28] M. J. Brewer and N. C. Elliott, “Biological control of cereal aphids in north america and mediating effects of host plant and habitat manipulations,” Annu. Rev. Entomol., vol. 49, pp. 219–242, 2004, doi: 10.1146/ANNUREV.ENTO.49.061802.123149.
[29] N. Desneux, A. Decourtye, and J. M. Delpuech, “The Sublethal Effects of Pesticides on Beneficial Arthropods,” http://dx.doi.org/10.1146/annurev.ento.52.110405.091440, vol. 52, pp. 81–106, Dec. 2006, doi: 10.1146/ANNUREV.ENTO.52.110405.091440.
[30] A. Decourtye, M. Henry, and N. Desneux, “Overhaul pesticide testing on bees,” Nat. 2013 4977448, vol. 497, no. 7448, pp. 188–188, May 2013, doi: 10.1038/497188a.
[31] N. Desneux, M. H. Pham-Delègue, and L. Kaiser, “Effects of sub-lethal and lethal doses of lambda-cyhalothrin on oviposition experience and host-searching behaviour of a parasitic wasp, Aphidius ervi,” Pest Manag. Sci., vol. 60, no. 4, pp. 381–389, Apr. 2004, doi: 10.1002/PS.822.
[32] N. Desneux, E. Wajnberg, X. Fauvergue, S. Privet, and L. Kaiser, “Oviposition behaviour and patch-time allocation in two aphid parasitoids exposed to deltamethrin residues,” Entomol. Exp. Appl., vol. 112, no. 3, pp. 227–235, 2004, doi: 10.1111/j.0013-8703.2004.00198.x.
[33] A. Biondi, O. Campolo, N. Desneux, G. Siscaro, V. Palmeri, and L. Zappalà, “Life stage-dependent susceptibility of Aphytis melinus DeBach (Hymenoptera: Aphelinidae) to two pesticides commonly used in citrus orchards,” Chemosphere, vol. 128, pp. 142–147, Jun. 2015, doi: 10.1016/J.CHEMOSPHERE.2015.01.034.
[34] N. A. Jam and M. Saber, “Sublethal effects of imidacloprid and pymetrozine on the functional response of the aphid parasitoid, Lysiphlebus fabarum,” Entomol. Gen., vol. 38, no. 2, pp. 173–190, 2018, doi: 10.1127/entomologia/2018/0734.
[35] A. A. A. H. Mohammed et al., “Impact of imidacloprid and natural enemies on cereal aphids: Integration or ecosystem service disruption?,” Entomol. Gen., vol. 37, no. 1, pp. 47–61, 2018, doi: 10.1127/entomologia/2017/0471.
[36] P. Liang, Y. A. Tian, A. Biondi, N. Desneux, and X. W. Gao, “Short-term and transgenerational effects of the neonicotinoid nitenpyram on susceptibility to insecticides in two whitefly species,” Ecotoxicology, vol. 21, no. 7, pp. 1889–1898, Oct. 2012, doi: 10.1007/S10646-012-0922-3.
[37] L. Xiaolan, T. Qiuling, L. Yongdan, R. Campos Mateus, L. Pei, and G. Xiwu, “Widespread resistance of the aphid Myzus persicae to pirimicarb across China, and insights on ace2 mutation frequency in this species,” Entomol. Gen., vol. 36, no. 4, pp. 285–299, 2017, doi: 10.1127/ENTOMOLOGIA/2017/0480.
[38] R. Weisz, S. Fleischer, and Z. Smilowitz, “Site-Specific Integrated Pest Management for High Value Crops: Sample Units for Map Generation Using the Colorado Potato Beetle (Coleoptera: Chrysomelidae) as a Model System,” J. Econ. Entomol., vol. 88, no. 5, pp. 1069–1080, Oct. 1995, doi: 10.1093/JEE/88.5.1069.
[39] L. Winder, J. N. Perry, and J. M. Holland, “The spatial and temporal distribution of the grain aphid Sitobion avenae in winter wheat,” Entomol. Exp. Appl., vol. 93, no. 3, pp. 277–290, 1999, doi: 10.1046/J.1570-7458.1999.00588.X.
[40] C. L. Young and M. G. Wright, “Seasonal and spatial distribution of banana aphid, Pentalonia nigronervosa (Hemiptera: Aphididae), in banana plantations on oahu,” Proc Hawaiian Entomol Soc, vol. 37, no. May, pp. 73–80, 2005, [Online]. Available: http://hdl.handle.net/10125/85.
[41] N. Desneux, R. J. O’neil, H. O. Jung, and S. Yoo, “Suppression of Population Growth of the Soybean Aphid, Aphis glycines Matsumura, by Predators: The Identification of a Key Predator and the Effects of Prey Dispersion, Predator Abundance, and Temperature,” Environ. Entomol., vol. 35, no. 5, pp. 1342–1349, Oct. 2006, doi: 10.1093/EE/35.5.1342.
[42] N. Desneux, J. M. Rabasse, Y. Ballanger, and L. Kaiser, “Parasitism of canola aphids in France in autumn,” J. Pest Sci. 2006 792, vol. 79, no. 2, pp. 95–102, Feb. 2006, doi: 10.1007/S10340-006-0121-1.
[43] M. Janković et al., “Functional role of different habitat types at local and landscape scales for aphids and their natural enemies,” J. Pest Sci. (2004)., vol. 90, no. 1, pp. 261–273, 2017, doi: 10.1007/s10340-016-0744-9.
[44] P. Han, C. Y. Niu, and N. Desneux, “Identification of Top-Down Forces Regulating Cotton Aphid Population Growth in Transgenic Bt Cotton in Central China,” PLoS One, vol. 9, no. 8, p. e102980, Aug. 2014, doi: 10.1371/JOURNAL.PONE.0102980.
[45] F. Yang et al., “Species composition and seasonal dynamics of aphid parasitoids and hyperparasitoids in wheat fields in northern China,” Sci. Reports 2017 71, vol. 7, no. 1, pp. 1–9, Oct. 2017, doi: 10.1038/s41598-017-14441-6.
[46] C. C. Jaworski et al., “Varying the spatial arrangement of synthetic herbivore-induced plant volatiles and companion plants to improve conservation biological control,” J. Appl. Ecol., vol. 56, no. 5, pp. 1176–1188, May 2019, doi: 10.1111/1365-2664.13353.
[47] J. Anwar et al., “Assessment of adaptability and stability of grain yield in bread wheat genotypes under different sowing times in Punjab,” Pakistan J. Bot., vol. 43, no. 4, pp. 1985–1993, 2011.
[48] H. S, K. A, M. A, F. S, and T. A, “Interference and economic threshold level of little seed canary grass in wheat under different sowing times,” Environ. Sci. Pollut. Res. Int., vol. 22, no. 1, pp. 441–449, Aug. 2015, doi: 10.1007/S11356-014-3304-Y.
[49] A. A. Khakwani, M. D. Dennett, M. Munir, and M. Abid, “Growth and yield response of wheat varieties to water stress at booting and anthesis stages of development,” Pakistan J. Bot., vol. 44, no. 3, pp. 879–886, 2012.
[50] M. S. Wains, M. W. Jamil, M. A. Ali, M. Hussain, and J. Anwar, “Germplasm screening and incorporation of aphid resistance in bread wheat (Triticum aestivum L.),” J. Anim. Plant Sci., vol. 24, no. 3, pp. 919–925, 2014.
[51] I.-H. Akhtar and . A. K., “Impact of Plant Phenology and Coccinellid Predators on the Population Dynamic of Rose Aphid Macrosiphum rosaeiformis Das (Aphididae: Homoptera) on Rose,” Asian J. Plant Sci., vol. 2, no. 1, pp. 119–122, Dec. 2002, doi: 10.3923/AJPS.2003.119.122.
[52] F. S. Murungu and T. Madanzi, “Seed priming, genotype and sowing date effects on emergence, growth and yield of wheat in a tropical low altitude area of Zimbabwe,” African J. Agric. Res., vol. 5, no. 17, pp. 2341–2349, 2010.
[53] R. Patterson, C. C. Extension, R. Ramirez, and E. E. Specialist, “Aphid Natural Enemies and Biological Control,” no. March, p. 5, 2016.
[54] C. A. Triplehorn, N. F. Johnson, and D. J. (Donald J. Borror, “Borror and DeLong’s introduction to the study of insects,” p. 864, 2005.


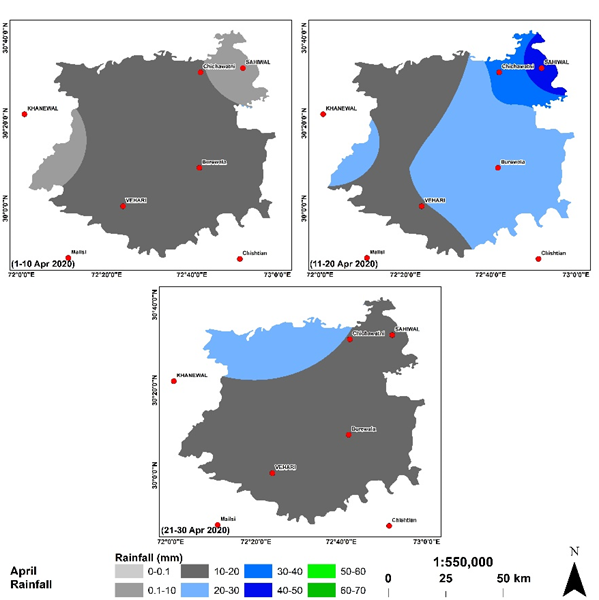 Figure 6. Spatial distribution of rainfall intensity during the month of April 2020.
Figure 6. Spatial distribution of rainfall intensity during the month of April 2020.


















