Photocatalytic Degradation of Deltamethrin in Drinking Water Under Visible Light by Using Zno and Tio2
Keywords:
Deltamethrin, Photo catalytic degradation, ZnO, TiO2, Oxidizing agentsAbstract
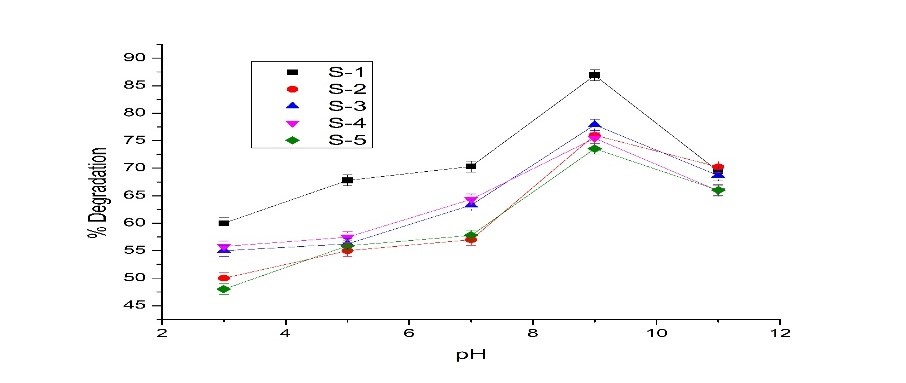
The use of deltamethrin is increasing due to its high demand in agriculture. However, it is toxic to both surface and groundwater. Agriculture plays a crucial role in the economy of any major nation. This study aims to enhance pesticide degradation by using specially designed catalysts optimized for visible light exposure. The key innovation lies in the customized catalyst design, which improves photocatalytic efficiency while offering a cost-effective and environmentally friendly approach. Various factors affecting degradation, including adsorbent quantity, pH, contact time, and initial concentration, were analyzed. The reactor consists of a 6-watt (380 nm) visible light lamp and a stirrer to ensure uniform mixing of the sample. Photocatalysts ZnO and TiO₂, in concentrations ranging from 0.1 to 3.0 g/L, were used to generate oxidizing agents. Under visible light, the impact of these factors on the degradation of different pesticide solutions was examined. The optimal doses were found to be 1.5 g/L for ZnO and 0.1 g/L for TiO₂. ZnO achieved a degradation rate of 96.3%, while TiO₂ slightly outperformed it with a rate of 96.34%. The study also investigated the effect of pH variations on deltamethrin degradation, revealing stronger degradation in alkaline conditions. Additionally, TiO₂ effectively reduced the COD value, demonstrating its superior efficiency in pesticide breakdown.
References
S. Fourmentin, G. Crini, and E. Lichtfouse, Eds., “Cyclodextrin Fundamentals, Reactivity and Analysis,” Environ. Chem. a Sustain. World, vol. 16, 2018, doi: 10.1007/978-3-319-76159-6.
P. Prashar and S. Shah, “Impact of Fertilizers and Pesticides on Soil Microflora in Agriculture,” Sustain. Agric. Rev., pp. 331–361, 2016, doi: 10.1007/978-3-319-26777-7_8.
I. H. Khuda Bakhsh, Nameem Ahmad, Shanza Tabasum, Sarfraz Hassan, “Health hazards and adoption of personal protective equipment during cotton harvesting in Pakistan,” Sci. Total Environ., vol. 598, pp. 1058–1064, 2017, doi: https://doi.org/10.1016/j.scitotenv.2017.04.043.
M. R. W. Jeffrey Farner Budarz, Ellen M. Cooper, Courtney Gardner, Emina Hodzic, P. Lee Ferguson, Claudia K. Gunsch, “Chlorpyrifos degradation via photoreactive TiO2 nanoparticles: Assessing the impact of a multi-component degradation scenario,” J. Hazard. Mater., vol. 372, pp. 61–68, 2019, doi: https://doi.org/10.1016/j.jhazmat.2017.12.028.
Ganeshwade R. M., “Biochemical changes induced by dimethoate (Rogor 30% EC) in the gills of fresh water fish Puntius ticto (Hamilton),” J. Ecol. Nat. Environ., vol. 4, no. 7, Apr. 2012, doi: 10.5897/JENE11.134.
A. Marican and E. F. Durán-Lara, “A review on pesticide removal through different processes,” Environ. Sci. Pollut. Res. 2017 253, vol. 25, no. 3, pp. 2051–2064, Nov. 2017, doi: 10.1007/S11356-017-0796-2.
J. V. Franck Delval, Grégorio Crini, “Removal of organic pollutants from aqueous solutions by adsorbents prepared from an agroalimentary by-product,” Bioresour. Technol., vol. 97, no. 16, pp. 2173–2181, 2006, doi: https://doi.org/10.1016/j.biortech.2005.09.022.
A. K. Jain, V. K. Gupta, A. Bhatnagar, and Suhas, “A Comparative Study of Adsorbents Prepared from Industrial Wastes for Removal of Dyes,” Sep. Sci. Technol., vol. 38, no. 2, pp. 463–481, 2003, doi: 10.1081/SS-120016585.
S. V. Vinod Kumar Gupta, Rajeev Jain, “Removal of Reactofix golden yellow 3 RFN from aqueous solution using wheat husk—An agricultural waste,” J. Hazard. Mater., vol. 142, no. 1–2, pp. 443–448, 2007, doi: https://doi.org/10.1016/j.jhazmat.2006.08.048.
V. K. S. Vinod Kumar Gupta, Imran Ali, “Defluoridation of wastewaters using waste carbon slurry,” Water Res., vol. 41, no. 15, pp. 3307–3316, 2007, doi: https://doi.org/10.1016/j.watres.2007.04.029.
A. N. V.K. Gupta, Bina Gupta, Arshi Rastogi, Shilpi Agarwal, “A comparative investigation on adsorption performances of mesoporous activated carbon prepared from waste rubber tire and activated carbon for a hazardous azo dye—Acid Blue 113,” J. Hazard. Mater., vol. 186, no. 1, pp. 891–901, 2011, doi: https://doi.org/10.1016/j.jhazmat.2010.11.091.
T. A. Saleh and V. K. Gupta, “Column with CNT/magnesium oxide composite for lead(II) removal from water,” Environ. Sci. Pollut. Res., vol. 19, no. 4, pp. 1224–1228, May 2012, doi: 10.1007/S11356-011-0670-6/METRICS.
A. K. Jain, V. K. Gupta, S. Jain, and Suhas, “Removal of Chlorophenols Using Industrial Wastes,” Environ. Sci. Technol., vol. 38, no. 4, pp. 1195–1200, Feb. 2004, doi: 10.1021/ES034412U.
M. M. Mubeena Akhtar, Shahid Iqbal, M.I. Bhanger, Muhammad Zia-Ul-Haq, “Sorption of organophosphorous pesticides onto chickpea husk from aqueous solutions,” Colloids Surfaces B Biointerfaces, vol. 69, no. 1, pp. 63–70, 2009, doi: https://doi.org/10.1016/j.colsurfb.2008.10.019.
A. N. Vinod K. Gupta, Arshi Rastogi, “Biosorption of nickel onto treated alga (Oedogonium hatei): Application of isotherm and kinetic models,” J. Colloid Interface Sci., vol. 342, no. 2, pp. 533–539, 2010, doi: https://doi.org/10.1016/j.jcis.2009.10.074.
G. S. S. Karthikeyan, V.K. Gupta, R. Boopathy, A. Titus, “A new approach for the degradation of high concentration of aromatic amine by heterocatalytic Fenton oxidation: Kinetic and spectroscopic studies,” J. Mol. Liq., vol. 173, pp. 153–163, 2012, doi: https://doi.org/10.1016/j.molliq.2012.06.022.
G. N. Simón Navarro, José Fenoll, Nuria Vela, Encarnación Ruiz, “Removal of ten pesticides from leaching water at pilot plant scale by photo-Fenton treatment,” Chem. Eng. J., vol. 167, no. 1, pp. 42–49, 2011, doi: https://doi.org/10.1016/j.cej.2010.11.105.
S. M. R. M.M. Ballesteros Martín, J.A. Sánchez Pérez, J.L. Casas López, I. Oller, “Degradation of a four-pesticide mixture by combined photo-Fenton and biological oxidation,” Water Res., vol. 43, no. 3, pp. 653–660, 2009, doi: https://doi.org/10.1016/j.watres.2008.11.020.
J. P. Farré, María José, Doménech, Xavier, “Assessment of photo-Fenton and biological treatment coupling for Diuron and Linuron removal from water,” Water Res., vol. 14, no. 13, pp. 2533–2540, 2006, doi: https://doi.org/10.1016/j.watres.2006.04.034.
D. W. Lhomme, Ludovic, Brosillon, Stephan, “Photocatalytic degradation of a triazole pesticide, cyproconazole, in water,” J. Photochem. Photobiol. A Chem., vol. 188, no. 1, pp. 34–42, 2007, doi: https://doi.org/10.1016/j.jphotochem.2006.11.015.
J. L. Casas López, A. Cabrera Reina, E. Ortega Gómez, M. M. Ballesteros Martín, S. Malato Rodríguez, and J. A. Sánchez Pérez, “Integration of Solar Photocatalysis and Membrane Bioreactor for Pesticides Degradation,” Sep. Sci. Technol., vol. 45, no. 11, pp. 1571–1578, Jan. 2010, doi: 10.1080/01496395.2010.487465.
J. S. J. Su-Gyeong Kim, Love Kumar Dhandole, Young-Seok Seo, Hee-Suk Chung, Weon-Sik Chae, Min Cho, “Active composite photocatalyst synthesized from inactive Rh & Sb doped TiO2 nanorods: Enhanced degradation of organic pollutants & antibacterial activity under visible light irradiation,” Appl. Catal. A Gen., vol. 564, pp. 43–55, 2018, doi: Applied Catalysis A: General.
P. P. Emmanouil S. Karafas, Manolis N. Romanias, Vassileios Stefanopoulos, Vassilios Binas, Apostolos Zachopoulos, George Kiriakidis, “Effect of metal doped and co-doped TiO2 photocatalysts oriented to degrade indoor/outdoor pollutants for air quality improvement. A kinetic and product study using acetaldehyde as probe molecule,” J. Photochem. Photobiol. A Chem., vol. 371, pp. 255–263, 2019, doi: https://doi.org/10.1016/j.jphotochem.2018.11.023.
A. Y. Sana Ahmad, “Photocatalytic degradation of deltamethrin by using Cu/TiO2/bentonite composite,” Arab. J. Chem., vol. 13, no. 11, pp. 8481–8488, 2020, doi: https://doi.org/10.1016/j.arabjc.2020.07.019.
A. Saljooqi, T. Shamspur, and A. Mostafavi, “Synthesis and photocatalytic activity of porous ZnO stabilized by TiO2 and Fe3O4 nanoparticles: investigation of pesticide degradation reaction in water treatment,” Environ. Sci. Pollut. Res., vol. 28, no. 8, pp. 9146–9156, Feb. 2021, doi: 10.1007/S11356-020-11122-2/METRICS.
L. R. M. Premalatha, N., “Surfactant modified ZnO–Bi2O3 nanocomposite for degradation of lambda- cyhalothrin pesticide in visible light: A study of reaction kinetics and intermediates,” J. Environ. Manage., vol. 246, pp. 259–266, 2019, doi: https://doi.org/10.1016/j.jenvman.2019.05.155.
A. P. T. Abhishek Sraw, Taranjeet Kaur, Yamini Pandey, Amit Sobti, Ravinder K. Wanchoo, “Fixed bed recirculation type photocatalytic reactor with TiO2 immobilized clay beads for the degradation of pesticide polluted water,” J. Environ. Chem. Eng., vol. 6, no. 6, pp. 7035–7043, 2018, doi: https://doi.org/10.1016/j.jece.2018.10.062.
Y. Abdel-Mobdy and E. Abdel-Rahim, “Toxicological Influences of Lambda Cyhalothrin and Evaluation of the Toxicity Ameliorative Effect of Pomegranate in Albino Rats,” Glob. Vet., vol. 14, no. 6, pp. 913–921, 2015, doi: 10.5829/idosi.gv.2015.14.06.95144.
N. A. Y. Yadollah Abdollahi, Abdul Halim Abdullah, Zulkarnain Zainal, “Photocatalytic Degradation of p-Cresol by Zinc Oxide under UV Irradiation,” Int. J. Mol. Sci, vol. 13, no. 1, pp. 302–315, 2012, doi: ttps://doi.org/10.3390/ijms13010302.
Downloads
Published
How to Cite
Issue
Section
License
Copyright (c) 2025 50SEA

This work is licensed under a Creative Commons Attribution 4.0 International License.




















